Home /
Expert Answers /
Chemistry /
what-values-from-question-1-should-i-use-in-my-calculation-for-question-2-also-where-does-the-value-pa811
(Solved): what values from question 1 should I use in my calculation for question 2? also where does the value ...
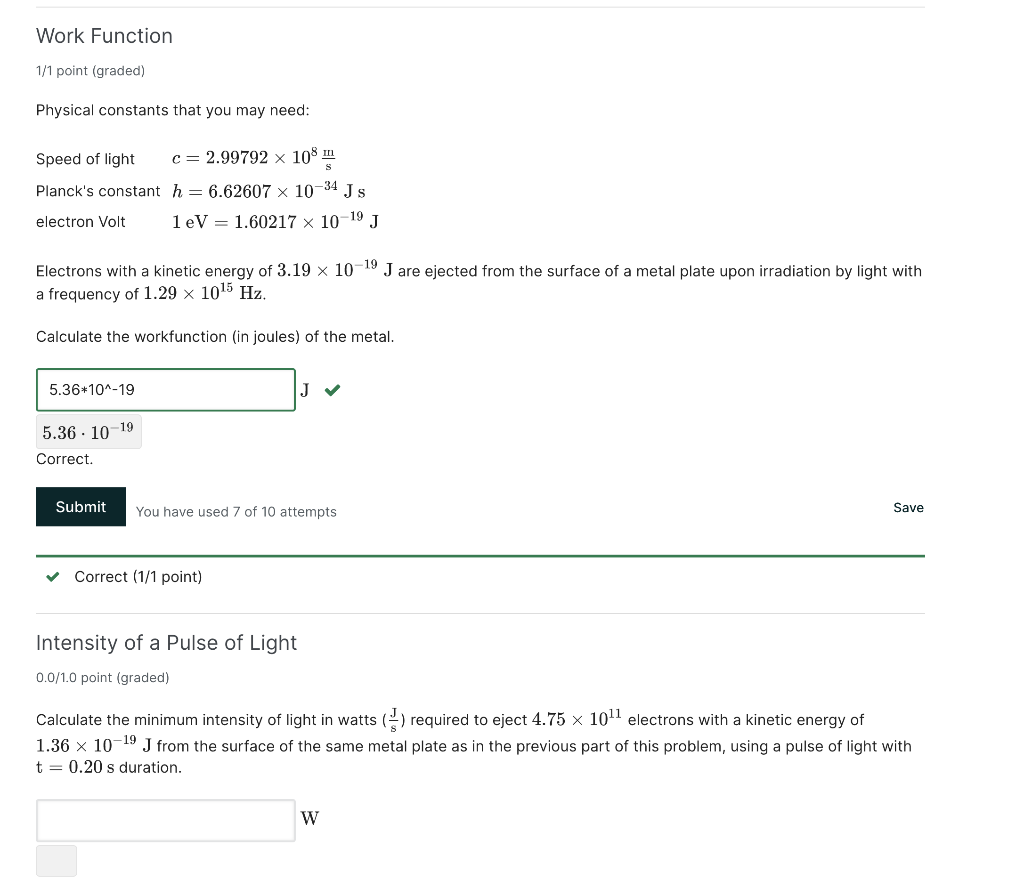
Work Function 1/1 point (graded) Physical constants that you may need: Speed of light Planck's constant electron Volt Electrons with a kinetic energy of are ejected from the surface of a metal plate upon irradiation by light with a frequency of . Calculate the workfunction (in joules) of the metal. Correct. You have used 7 of 10 attempts Save Correct ( point) Intensity of a Pulse of Light point (graded) Calculate the minimum intensity of light in watts required to eject electrons with a kinetic energy of from the surface of the same metal plate as in the previous part of this problem, using a pulse of light with duration. W
Expert Answer
Here we will use the workfunction from question 1 which is 5.36*10^-19.here the time will be used in power calculations.i will show the use below here