Home /
Expert Answers /
Chemical Engineering /
solve-part-b-please-in-your-car-the-catalytic-converter-oxidizes-carbon-monoxide-mathrm-co-pa119
(Solved): SOLVE PART B, please In your car, the catalytic converter oxidizes carbon monoxide \( (\mathrm{CO ...
SOLVE PART B, please
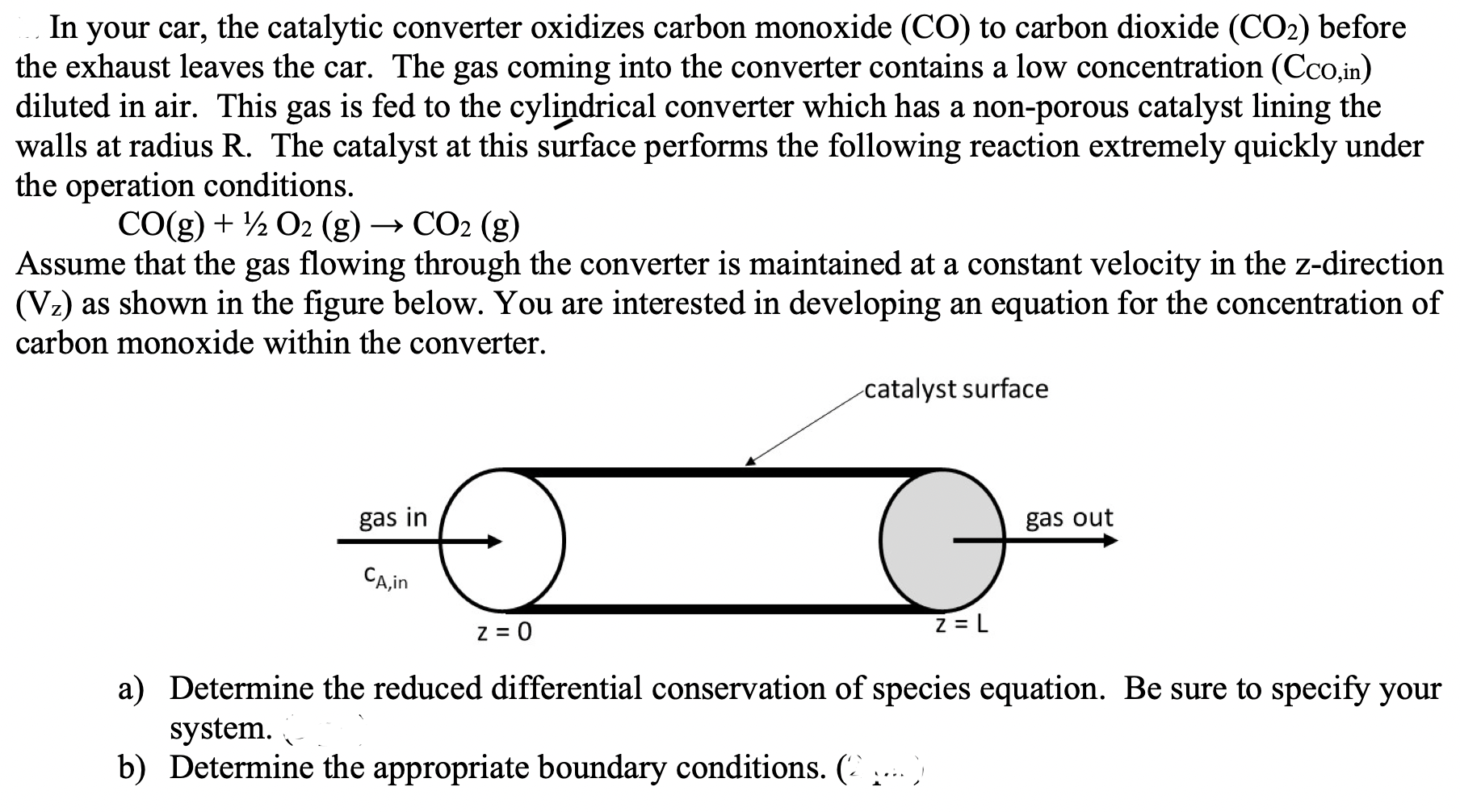
In your car, the catalytic converter oxidizes carbon monoxide \( (\mathrm{CO}) \) to carbon dioxide \( \left(\mathrm{CO}_{2}\right) \) before the exhaust leaves the car. The gas coming into the converter contains a low concentration \( \left(\mathrm{C}_{\mathrm{CO}, \text { in }}\right) \) diluted in air. This gas is fed to the cylindrical converter which has a non-porous catalyst lining the walls at radius \( \mathrm{R} \). The catalyst at this surface performs the following reaction extremely quickly under the operation conditions. \[ \mathrm{CO}(\mathrm{g})+1 / 2 \mathrm{O}_{2}(\mathrm{~g}) \rightarrow \mathrm{CO}_{2}(\mathrm{~g}) \] Assume that the gas flowing through the converter is maintained at a constant velocity in the z-direction \( \left(\mathrm{V}_{z}\right) \) as shown in the figure below. You are interested in developing an equation for the concentration of carbon monoxide within the converter. a) Determine the reduced differential conservation of species equation. Be sure to specify your system. b) Determine the appropriate boundary conditions. \( (\ldots, \ldots \),
Expert Answer
Solution: Boundary conditions for the given above system is, The first boundary condition, equation (1), recognizes that as soon as the f