Home /
Expert Answers /
Chemistry /
propane-gas-c3h8-reacts-with-oxygen-to-produce-water-and-carbon-dioxide-c3h8-g-5o2-pa816
(Solved): Propane gas, C3H8, reacts with oxygen to produce water and carbon dioxide. C3H8(g)+5O2 ...
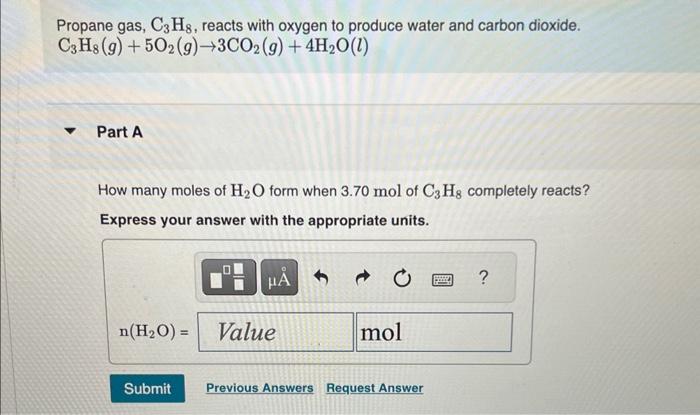
Propane gas, , reacts with oxygen to produce water and carbon dioxide. Part A How many moles of form when of completely reacts? Express your answer with the appropriate units.
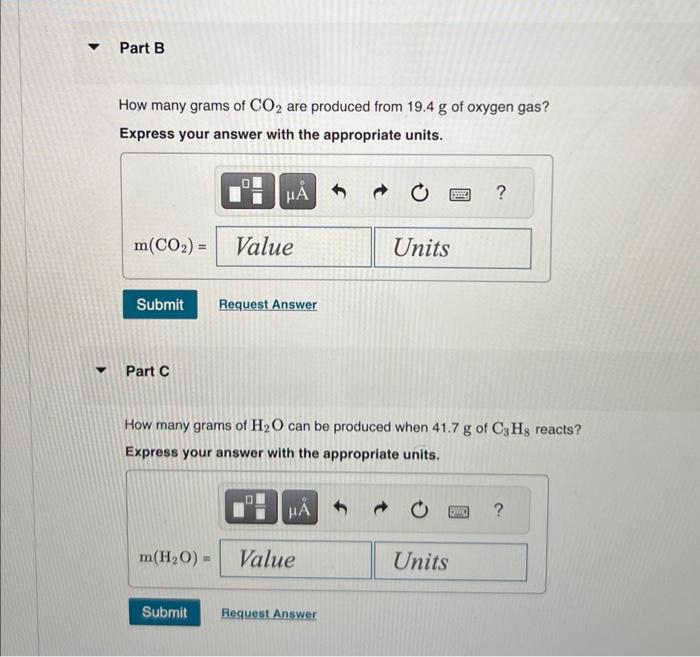
How many grams of are produced from of oxygen gas? Express your answer with the appropriate units. Part C How many grams of can be produced when of reacts? Express your answer with the appropriate units.