Home /
Expert Answers /
Chemistry /
please-fill-in-the-blanks-for-the-given-pictures-bellow-part-a-data-and-calculations-measurement-or-pa353
(Solved): please fill in the blanks for the given pictures bellow Part A data and calculations Measurement or ...
please fill in the blanks for the given pictures bellow
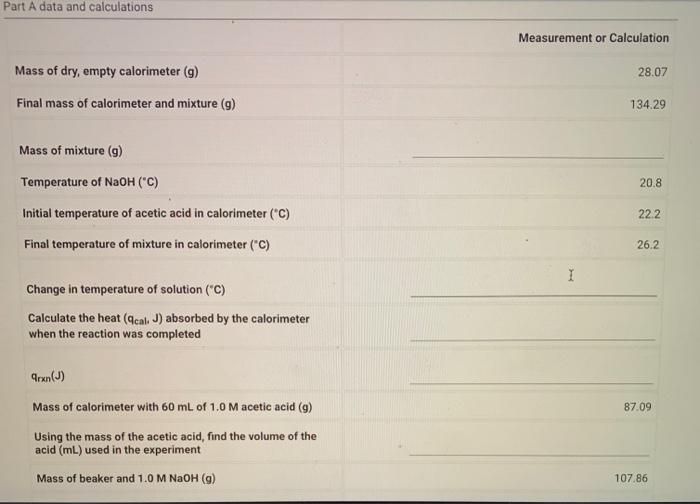

Part A data and calculations Measurement or Calculation Mass of dry, empty calorimeter Final mass of calorimeter and mixture ( ) Mass of mixture (g) Temperature of Initial temperature of acetic acid in calorimeter ("C) Final temperature of mixture in calorimeter ("C) Change in temperature of solution ("C) Calculate the heat absorbed by the calorimeter when the reaction was completed Mass of calorimeter with of acetic acid 87.09 Using the mass of the acetic acid, find the volume of the acid used in the experiment Mass of beaker and 1.0
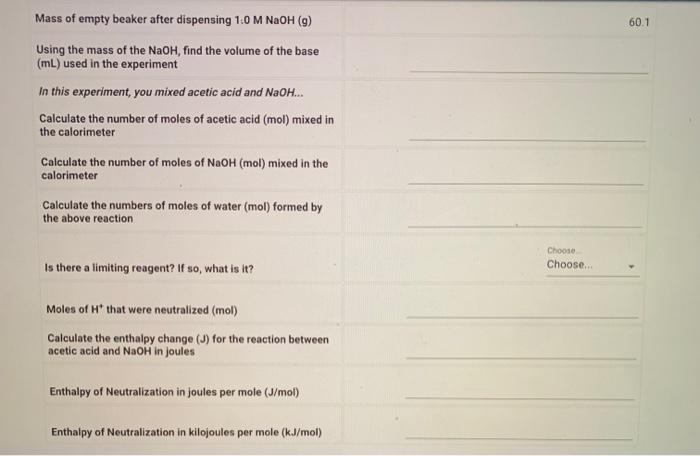
Mass of empty beaker after dispensing 1:0 M NaOH (g) Using the mass of the , find the volume of the base used in the experiment In this experiment, you mixed acetic acid and . Calculate the number of moles of acetic acid (mol) mixed in the calorimeter Calculate the number of moles of (mol) mixed in the calorimeter Calculate the numbers of moles of water (mol) formed by the above reaction Is there a limiting reagent? If so, what is it? Moles of that were neutralized (mol) Calculate the enthalpy change for the reaction between acetic acid and in joules Enthalpy of Neutralization in joules per mole Enthalpy of Neutralization in kilojoules per mole
Expert Answer
To calculate the heat absorbed by the calorimeter, we can use the following formula:q = mc?Twhere q is the heat absorbed, m is the mass of the substan