Home /
Expert Answers /
Chemistry /
help-the-solubility-product-constant-for-cobalt-ii-carbonate-is-8-0-times-10-13-sol-pa698
(Solved): help The Solubility Product Constant for cobalt(II) carbonate is \( 8.0 \times 10^{-13} \). Sol ...
help
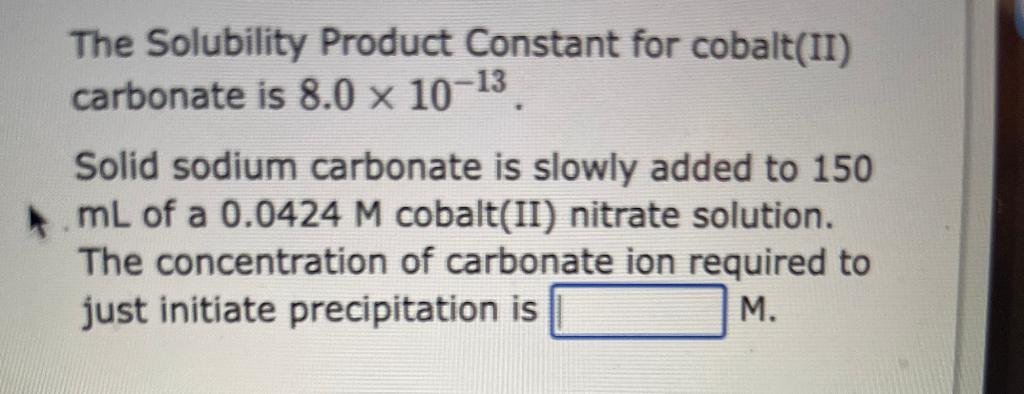
The Solubility Product Constant for cobalt(II) carbonate is \( 8.0 \times 10^{-13} \). Solid sodium carbonate is slowly added to 150 \( \mathrm{mL} \) of a \( 0.0424 \mathrm{M} \) cobalt(II) nitrate solution. The concentration of carbonate ion required to just initiate precipitation is M.
Solid manganese(II) nitrate is slowly added to \( 50.0 \mathrm{~mL} \) of a \( 0.0345 \mathrm{M} \) potassium hydroxide solution. The concentration of manganese(II) ion required to just initiate precipitation is M. (Assume that \[ \left.K_{\mathrm{sp}}\left(\mathrm{Mn}(\mathrm{OH})_{2}\right)=4.6 \times 10^{-14} .\right) \]