Home /
Expert Answers /
Chemistry /
determine-the-mass-of-a-solution-containing-5-00-grams-of-mathrm-kcl-in-1-pa166
(Solved): Determine the \( \% \) mass of a solution containing \( 5.00 \) grams of \( \mathrm{KCl} \) in \( 1 ...
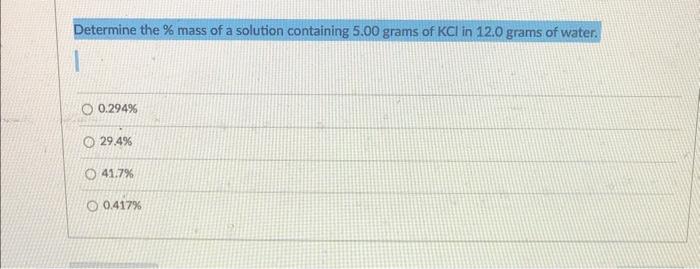

Determine the \( \% \) mass of a solution containing \( 5.00 \) grams of \( \mathrm{KCl} \) in \( 12.0 \) grams of water. \( 0.294 \% \) \( 29.4 \% \) \( 41.7 \% \) \( 0.417 \% \)
What is the volume of \( 0.255 \) mole of helium with a pressure of \( 7.48 \mathrm{~atm} \) at 280 . K? \[ (R=0.0821 \mathrm{~L} \cdot \mathrm{atm} / \mathrm{mol} \cdot \mathrm{K}) \] A. \( 1.55 \mathrm{~L} \) B. \( 0.784 \mathrm{~L} \) C. \( 0.00842 \mathrm{~L} \) D. none of the above
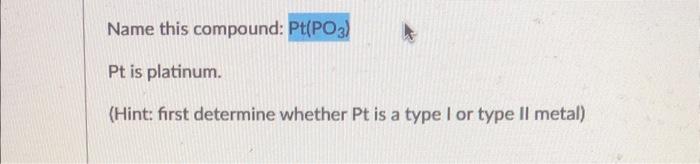
Name this compound: Pt is platinum. (Hint: first determine whether Pt is a type I or type II metal)