Home /
Expert Answers /
Chemistry /
consider-the-following-energy-diagram-showing-two-posslble-and-competing-pathways-for-the-reaction-pa391
(Solved): Consider the following energy diagram showing two posslble and competing pathways for the reaction ...
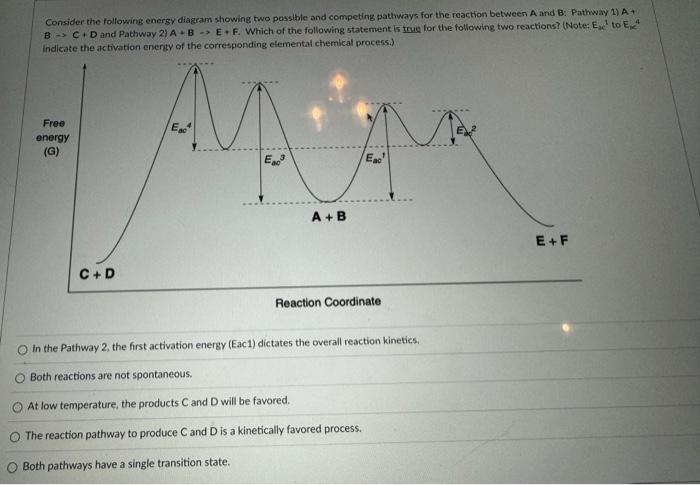
Consider the following energy diagram showing two posslble and competing pathways for the reaction between A and B: Pathway 1) A and Pathway 2) . Which of the following statement is true for the following two reactions? (Note: to indicate the activation enersy of the corresponding elemental chemical process.) In the Pathway 2, the first activation energy (Eac1) dictates the overall reaction kinetics. Both reactions are not spontaneous. At low temperature, the products and will be favored. The reaction pathway to produce and is a kinetically favored process. Both pathways have a single transition state.