Home /
Expert Answers /
Chemistry /
compound-mathbf-w-mathrm-c-8-mathrm-h-17-mathrm-br-text-and-mathbf-x-ma-pa813
(Solved): Compound \( \mathbf{W}, \mathrm{C}_{8} \mathrm{H}_{17} \mathrm{Br}_{\text {, and }} \mathbf{X}, \ma ...
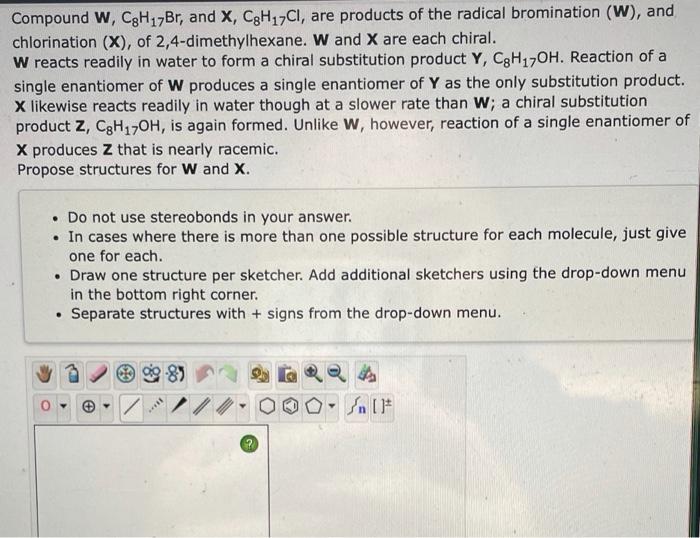
Compound \( \mathbf{W}, \mathrm{C}_{8} \mathrm{H}_{17} \mathrm{Br}_{\text {, and }} \mathbf{X}, \mathrm{C}_{8} \mathrm{H}_{17} \mathrm{Cl} \), are products of the radical bromination (W), and chlorination \( (\mathbf{X}) \), of 2,4-dimethylhexane. \( \mathbf{W} \) and \( \mathbf{X} \) are each chiral. W reacts readily in water to form a chiral substitution product \( \mathbf{Y}, \mathrm{C}_{8} \mathrm{H}_{17} \mathrm{OH} \). Reaction of a single enantiomer of \( \mathbf{W} \) produces a single enantiomer of \( \mathbf{Y} \) as the only substitution product. \( \mathbf{X} \) likewise reacts readily in water though at a slower rate than \( \mathbf{W} \); a chiral substitution product \( \mathbf{Z}, \mathrm{C}_{8} \mathrm{H}_{17} \mathrm{OH} \), is again formed. Unlike \( \mathbf{W} \), however, reaction of a single enantiomer of \( \mathbf{X} \) produces \( \mathbf{Z} \) that is nearly racemic. Propose structures for \( \mathbf{W} \) and \( \mathbf{X} \). - Do not use stereobonds in your answer. - In cases where there is more than one possible structure for each molecule, just give one for each. - Draw one structure per sketcher. Add additional sketchers using the drop-down menu in the bottom right corner. - Separate structures with + signs from the drop-down menu.