Home /
Expert Answers /
Chemistry /
blast-furnaces-extract-pure-iron-from-the-iron-iii-oxide-in-iron-ore-in-a-two-step-sequence-in-th-pa363
(Solved): Blast furnaces extract pure iron from the iron(III) oxide in iron ore in a two step sequence. In th ...
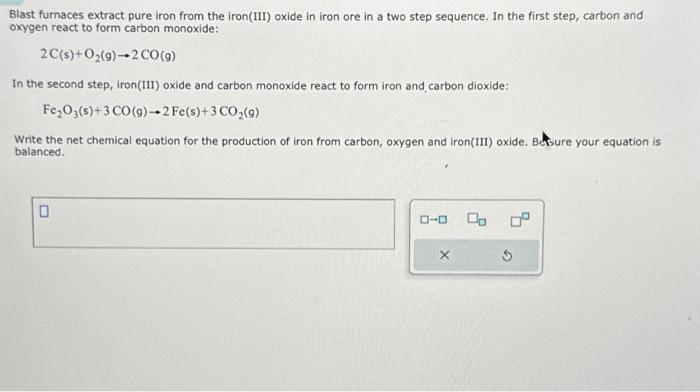
Blast furnaces extract pure iron from the iron(III) oxide in iron ore in a two step sequence. In the first step, carbon and oxygen react to form carbon monoxide: In the second step, iron(III) oxide and carbon monoxide react to form iron and carbon dioxide: Write the net chemical equation for the production of iron from carbon, oxygen and iron(III) oxide. Bure your equation is balanced.