Home /
Expert Answers /
Chemistry /
at-500c-cyclopropane-c3h6-rearranges-to-propene-ch3ch-ch2-the-reaction-is-f-pa398
(Solved): At 500C, cyclopropane (C3H6) rearranges to propene (CH3CH=CH2). The reaction is f ...

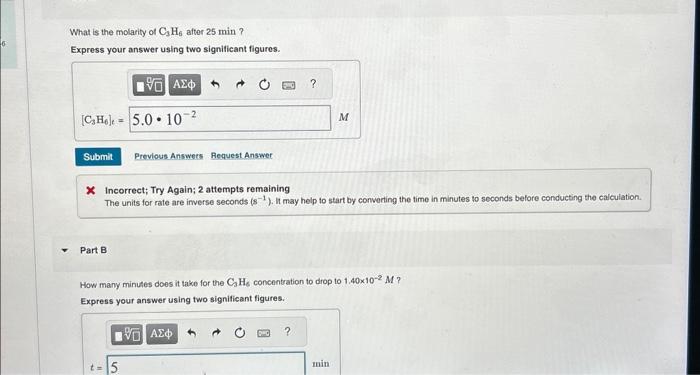

At , cyclopropane rearranges to propene . The reaction is first order, and the rate constant is . If the initial concentration of is .
What is the molarity of after ? Express your answer using two significant figures. X Incorrect; Try Again; 2 attempts remaining The units for rate are inverse seconds . It may help to start by converting the time in minutes to soconds belore conducting the calculation. Part B How many minutes does it take for the concentration to drop to ? Express your answer using two significant figures.
How many minutes does it take for of the to react? Express your answer using two significant figures.