Home /
Expert Answers /
Chemistry /
40-0-ml-of-0-200-m-h2nnh2-solution-was-titrated-with-100-ml-of-0-1-m-hno3-a-strong-acid-fill-in-t-pa317
(Solved): 40.0 mL of 0.200 M H2NNH2 solution was titrated with 100 mL of 0.1 M HNO3 (a strong acid). Fill in t ...
40.0 mL of 0.200 M H2NNH2 solution was titrated with 100 mL of 0.1 M HNO3 (a strong acid). Fill in the BCA table with the appropriate value to determine the moles of reactant and product after the reaction of acid and base
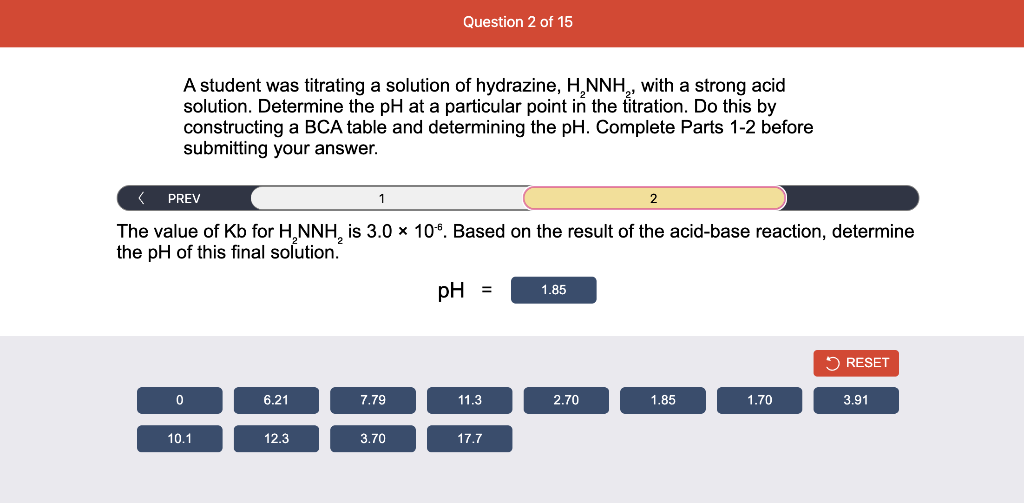
A student was titrating a solution of hydrazine, , with a strong acid solution. Determine the at a particular point in the titration. Do this by constructing a table and determining the . Complete Parts 1-2 before submitting your answer. of a solution was titrated with of (a strong acid). Fill in the BCA table with the appropriate value for each involved species to determine the moles of reactant and product after the reaction of the acid and base..
A student was titrating a solution of hydrazine, , with a strong acid solution. Determine the at a particular point in the titration. Do this by constructing a table and determining the . Complete Parts 1-2 before submitting your answer. The value of for is . Based on the result of the acid-base reaction, determine the of this final solution.