Home /
Expert Answers /
Chemistry /
1-draw-the-ketone-that-is-in-equilibrium-with-the-given-enol-below-during-tautomerization-draw-the-pa389
(Solved): 1. Draw the ketone that is in equilibrium with the given enol below during tautomerization; draw the ...
1. Draw the ketone that is in equilibrium with the given enol below during tautomerization; draw the enol that is in equilibrium with the given ketone or aldehyde (0.25 pt, 0.125 pt each)
2. Draw the kinetic and thermodynamic enolates formed when 3-methylbutan-2-one (aka methyl isopropyl ketone) is treated with a base (Na OCHs, for example. Please make sure to draw the oxide form, not the carbanion form. (0.75 pts, 0.25 pt each)
3. LDA (aka lithium disopropylamide) is a strong, nonnucleophilic base. It is therefore a very useful base in carbonyl chemistry and it can be freshly prepared by treating disopropylamine with n-butyllithium. Complete this reaction below. (0.75 pts, 0.25 pt each)
WILL UPVOTE
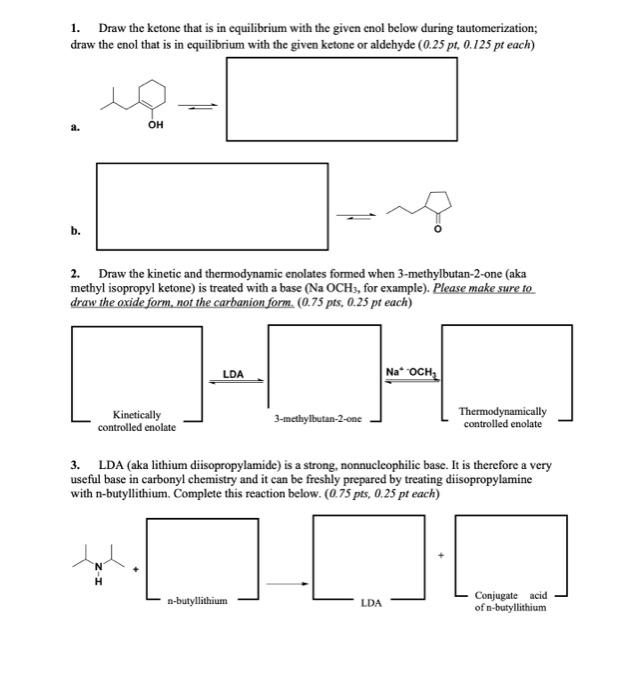
1. Draw the ketone that is in equilibrium with the given enol below during tautomerization; draw the enol that is in equilibrium with the given ketone or aldehyde \( (0.25 \mathrm{pt}, 0.125 \mathrm{pt} \) each \( ) \) a. b. 2. Draw the kinetic and thermodynamic enolates formed when 3-methylbutan-2-one (aka methyl isopropyl ketone) is treated with a base ( \( \mathrm{Na} \mathrm{OCH}_{3} \), for example). Please make sure to draw the oxide form. not the carbanion form. \( (0.75 \mathrm{pts}, 0.25 \mathrm{pt} \) each) 3. LDA (aka lithium disopropylamide) is a strong, nonnucleophilic base. It is therefore a very useful base in carbonyl chemistry and it can be freshly prepared by treating diisopropylamine with n-butyllithium. Complete this reaction below. \( (0.75 \mathrm{pts}, 0.25 \mathrm{pt} \) each \( ) \)