Home /
Expert Answers /
Chemistry /
1-below-is-the-titration-curve-for-carbonic-acid-h2co3-a-what-ph-range-or-ranges-is-carbonic-a-pa131
(Solved): 1. Below is the titration curve for carbonic acid H2CO3. a. What pH range or ranges is carbonic a ...
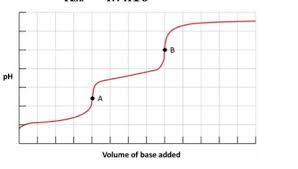
1. Below is the titration curve for carbonic acid H2CO3.
a. What pH range or ranges is carbonic acid an effective buffer at?
b. What are points A and B representing on this curve?
c. What are the predominate ionic forms of carbonic acid at point A on the titration curve?
d. What is the molar ratio of CO3 2- to HCO3 - at pH 9.5?
Volume of base added
Expert Answer
Answer: a. The pH range in which carbonic acid is an effective buffer is around pH 6 to 8. At this pH range, carbonic acid is mostly in its acid form